Today’s consumers demand high performance from the products they purchase such as apparel, home textiles and flooring products, so combating the undesirable effects of microbial growth is of utmost importance.
What are microbes?
Microbes are organisms that often cannot be seen by the naked eye, requiring the use of a microscope to be clearly seen. They include bacteria, algae, fungi, protozoa and micro-animals such as house dust mites.
Although these organisms are microscopic, they are present in abundance all around us.
On unprotected materials, their growth rate in optimal environmental conditions can be rapid, with some bacteria doubling every 20 minutes. Once growth takes hold, these microorganisms will cause unpleasant odors, staining and product degradation, reducing the useful life of untreated fibers, textiles and nonwovens.
What are antimicrobial treatments?
An antimicrobial treatment is an active agent that, when included in the manufacturing process of a product or material, works to inhibit the growth and even eliminate microorganisms from occurring within the material or product throughout its entire lifespan.
Antimicrobials may be referred to as either antibacterial or antifungal agents. Antibacterial treatments control the growth of bacteria, while antifungal treatments control the growth of fungi, mold and mildew.
How are antimicrobial treatments added to fibers?
Depending on the fiber type, diameter size and processing conditions, antimicrobials can be incorporated using several different methods. They can be added directly into a fiber or the fiber sheath during extrusion, or topically added after the fibers have been manufactured, either by polymer coating, spray application or by introducing into the spin finish.
For nonwoven products, if options are not available to treat the fibers before production, antimicrobial additives can be incorporated during the bonding or finishing processes. Knitted or woven textiles are typically treated using a pad application or by exhaust.
What are the benefits of adding antimicrobial treatments to fibers?
The potential for the proliferation of bacteria, mold and mildew exists wherever moisture is present.
Apparel retains moisture and nutrients from sweat, leading to the exponential growth of bacteria, along with their embarrassing odors.
Materials such as automotive upholstery, household carpets and rugs are under a constant barrage of dust, dirt and spills, creating optimal conditions for mold and mildew to flourish; the results are musty odors and undesirable staining.
Construction materials, commonly utilizing nonwoven components, are often exposed to environmental conditions that are more than favorable for microbial growth, leading to product deterioration and loss of integrity.
Finished goods treated with antimicrobial additives inhibit growth of bacteria, mold and mildew, which promises freshness, cleanliness and enhanced product durability. They provide an economical means of increasing value and performance while also offering a competitive edge in the market. In addition antimicrobial treatments extend the useful life of treated products, promoting environmental responsibility.
How is efficacy of antimicrobial-treated fibers measured?
Many test methods developed by organizations such as the American Association of Textile Chemists and Colorists (AATCC), the American Society for Testing and Materials (ASTM), the International Organization for Standardization (ISO) and the Japanese Industrial Standard (JIS) are available to evaluate antimicrobial performance.
Such standardized test methods are often developed for specific types of materials, end-uses or antimicrobial technologies, therefore choosing the correct test method is crucial.
Test methods to assess antimicrobial efficacy
Antimicrobial test methods are usually grouped into two general categories: qualitative and quantitative.
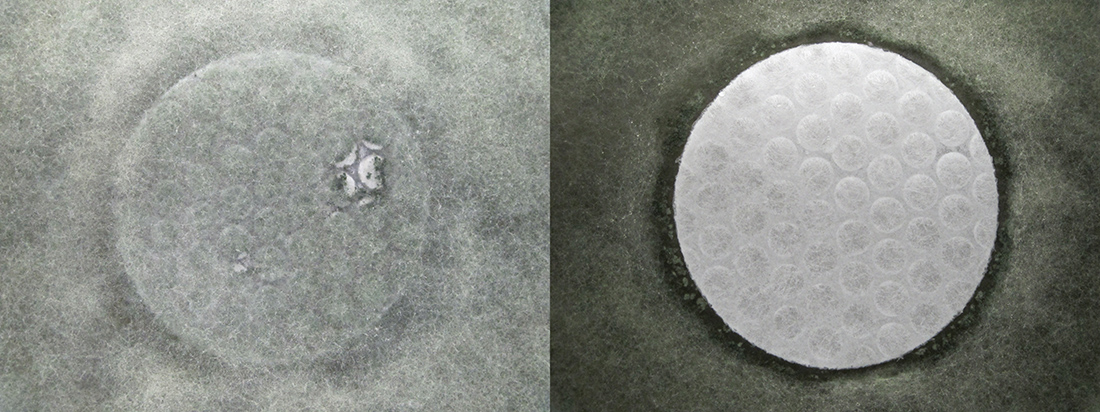
Qualitative tests such as the antibacterial AATCC Method 147 or the antifungal AATCC Method 30 and ASTM G21 are based on visual observations of microbial growth, with a microscope used to confirm an absence of macroscopic growth.
Such methods can utilize a growth-free zone approach, also known as zone-of-inhibition, to roughly estimate the antibacterial/antifungal activity of a material. However, there is not necessarily a correlation between a zone of inhibition and the activity of a product.
Such visual observation, as seen in Figure 1, make these test methods easy to understand for those without a scientific background. However, qualitative tests are not appropriate to measure activity of many types of antimicrobials.
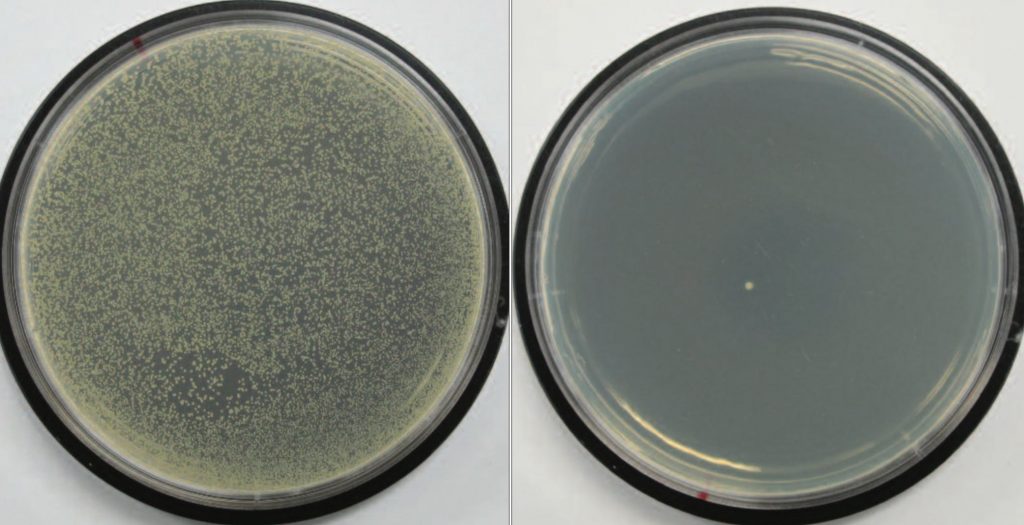
Quantitative tests, on the other hand, determine the degree of antimicrobial activity using a numeric value such as percent reduction or log reduction. Common antibacterial quantitative methods are ISO 20743, JIS L 1902, AATCC Method 100 and ASTM E2149.
With these methods a known number of bacteria are placed into contact with either a tightly wound bundle of fibers or the finished textile/nonwoven material. After a defined incubation period, the surviving bacteria are recovered and recounted as shown in Figure 2.
In addition to the most common qualitative and quantitative test methods mentioned above, certain industries may develop specific antimicrobial test criteria that the fiber or the resulting nonwoven or textile must meet. For example, most automotive manufacturers have designed their own testing specification to assess odor formation or susceptibility to mold and mildew growth.
Quantitative tests are more sensitive than qualitative methods and can be used for a wider range of antimicrobial products. They provide more relevant real-world test conditions and the quantitative measurement of performance is a more meaningful result than a zone of inhibition.
However, quantitative tests are much more expensive and time consuming to perform than qualitative tests. It is also imperative that an experienced laboratory is selected to conduct quantitative tests, as inexperienced or poorly trained microbiologists can lead to a wide variation of results.
Ultra-Fresh antimicrobial additives
Ultra-Fresh is a range of antimicrobial treatments developed by Thomson Research Associates (TRA), a company with more than 60 years of antimicrobial expertise.
Ultra-Fresh antimicrobials work continuously to control the growth of bacteria, fungi and algae that can lead to unpleasant smells, stains and degradation. This added protection assures treated goods remain clean and fresh for the useful life of the finished product.
Ultra-Fresh antimicrobial additives have a proven performance in the highlighted test methods, as well as many other standardized methods not discussed in this article. TRA’s state-of-the-art laboratories are equipped to perform thousands of these tests every year by rigorously trained microbiologists that are experts in their field.
TRA provides efficacy testing during the developmental phase of any Ultra-Fresh program and continues with complementary quality control testing of established programs to ensure treated products maintain a high level of performance.
In addition to laboratory testing TRA offers marketing support and regulatory assistance to help manufacturers navigate US Environmental Protection Agency (EPA) guidelines for claims surrounding antimicrobial-treated articles as per the Pesticide Registration Notice 2000-1.
Ultra-Fresh treatments have had to pass the most stringent testing procedures for safety and efficacy to meet international regulatory guidelines. These products are EPA registered, Biocidal Products Regulation (BPR) compliant, Oekotex listed and comply with all regulatory requirements of each country where they are sold.
Further, TRA has partnered with Bluesign in a joint initiative for the removal of harmful substances and practices from the textile manufacturing process.